UIHC joins study analyzing the immune response to fourth COVID-19 shot
University of Iowa Health Care is one of more than 20 locations participating in a vaccine study analyzing how the human body reacts to a second COVID-19 booster tailored to variants of the virus.
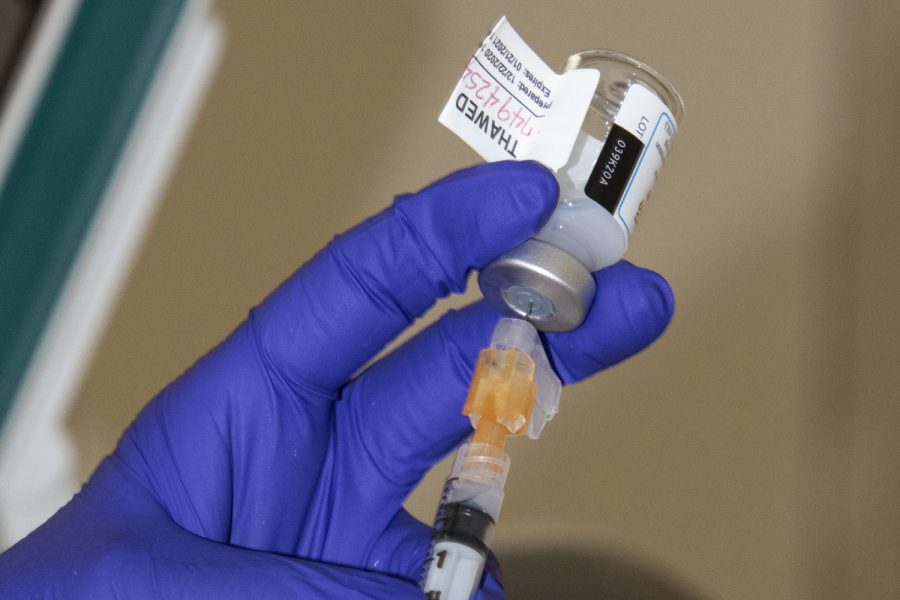
Moderna COVID-19 vaccine is drawn up on Tuesday, Dec. 22, 2020.
May 5, 2022
University of Iowa Health Care is participating in a vaccine trial studying the immune responses of individuals shortly after they have received their second booster vaccine against COVID-19.
The National Institute of Allergy and Infectious Diseases at the National Institutes of Health is sponsoring the trial, which is being conducted around the country at more than 20 sites.
Patricia Winokur, executive dean of the Carver College of Medicine and professor of internal medicine covering infectious diseases, said that doctors and scientists who have studied COVID-19 vaccines find that they are effective in the short term, but protection wears down quickly over time.
She said the virus has evolved into different variants of the original strain. Winokur said this means that to develop the most effective vaccine possible, researchers must better understand the response of the body.
“We’re trying to understand, very deeply, the immune response to these vaccines,” Winokur said. “By combining different variant vaccines, prototype vaccines, we’re hoping we will start to understand that really intricate immune response that occurs from the SARS-COVID vaccines as a whole.”
Variant vaccines, which are tailored towards variants of the virus, are manufactured in the same manner as the original vaccines against COVID-19, using mRNA, but that messenger RNA blueprint for the vaccines, Winokur said, has been altered.
“The variant vaccines do protect better against the variant strains – usually it’s about 13 times higher antibody protection,” Winokur said. “The one thing we’re still happy to see is that getting boosted with the original strain vaccine, the prototype vaccine, people have been protected from severe disease, even with the variant viruses that have been circulating.”
UI Health Care received funding and is participating in this trial because of its reputation, Winokur said. Vaccine patients have been very compliant and the UI has had a very good history of providing good data for previous vaccine trials as well, she said.
Winokur said that COVID-19 could become quite similar to influenza, with strains changing every single year and the need for an updated vaccination blueprint, like with the flu shot.
“We know, every year, flu changes its outer coat proteins as well, and we have to keep evolving our vaccines to those new variants that occur year-by-year to try and really get the best protection from those vaccines,” Winokur said. “It’s very possible that this may be the case for the coronaviruses as well.”